News Releases
New "plus" version of test adds a 3rd gene target to enhance detection of future SARS-CoV-2 variants
SUNNYVALE, Calif., May 12, 2022 /PRNewswire/ -- Cepheid today announced it has received Emergency Use Authorization (EUA) from the U.S. Food & Drug Administration (FDA) for Xpert® Xpress CoV-2 plus, a rapid molecular diagnostic test for qualitative detection of the virus that causes COVID-19.
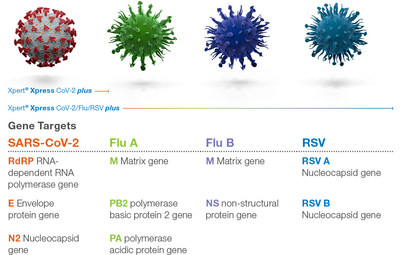
Viruses constantly change through mutation and these mutations can give rise to new variants with unique characteristics. Multiple variants of the virus that cause COVID-19 have been documented globally during the pandemic. Cepheid is proactively addressing this increasing genetic diversity by enhancing gene coverage. The new plus version of the test incorporates a 3rd conserved genetic target for SARS-CoV-2 detection to meet the challenge of future viral mutations and optimizes nucleocapsid gene probes to enable consistent virus detection.
Xpert Xpress CoV-2 plus joins Xpert® Xpress CoV-2/Flu/RSV plus and others in Cepheid's growing portfolio of PCRplus respiratory tests that deliver rapid, accurate, and actionable respiratory results. Xpert Xpress CoV-2/Flu/RSV plus continues to be the most appropriate product for when multiple viruses that cause influenza-like illnesses are circulating. Xpert Xpress CoV2 plus is authorized to be used on any individuals, including for screening those without symptoms or reasons to suspect COVID-19.1
Xpert Xpress CoV-2 plus is designed for use on any of Cepheid's over 40,000 GeneXpert® systems placed worldwide. The test can provide rapid on-demand detection of SARS-CoV-2 in as soon as 20 minutes for positive results.2
"From the beginning of the pandemic, we have been keenly focused on staying ahead of SARS-CoV-2 genetic drift and have designed our tests in anticipation of current and potential future variants." said David Persing, M.D., Ph.D., EVP, and Chief Scientific Officer. "The high sensitivity of this test is now especially important for recently announced Test-to-Treat initiatives, for which early detection is important for achieving the best clinical outcomes of antiviral therapies."
Xpert Xpress CoV-2 plus is expected to begin shipping to US customers in May.
Visit www.cepheid.com/coronavirus for more information, videos and package inserts.
1. PPA and NPA for asymptomatic specimens were calculated using anterior nasal swab specimens
2. With early assay termination for positives only; reporting of negatives in approximately 30 minutes
About Cepheid
Based in Sunnyvale, Calif., Cepheid is a leading molecular diagnostics company. Cepheid is dedicated to improving healthcare by developing, manufacturing, and marketing accurate yet easy-to-use molecular systems and tests. By automating highly complex and time-consuming manual procedures, the company's solutions deliver a better way for institutions of any size to perform sophisticated molecular diagnostic testing for organisms and genetic-based diseases. Through its strong molecular biology capabilities, the company is focusing on those applications where accurate, rapid, and actionable test results are needed most, such as managing infectious diseases and cancer. For more information, visit http://www.cepheid.com.
About Emergency Use Authorization Status
This product has not been FDA cleared or approved, but has been authorized by FDA under an EUA for use by authorized laboratories. This product has been authorized only for the detection of nucleic acid from SARS-CoV-2, not for any other viruses or pathogens. The emergency use of this product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.
For Cepheid Media Inquiries:
Darwa Peterson
darwa.peterson@cepheid.com
SOURCE Cepheid